|
Low atomic number materials are more efficient at scattering X-rays than higher atomic materials. Potts, Kouichi Tsuji, in Encyclopedia of Analytical Science (Third Edition), 2019 ED-XRF Using Polarized Excitation Oxford: Blackwell Scientific Publications.) (1993) Quantities, Units and Symbols in Physical Chemistry, 2nd edn. (Reprinted with permission from Mills I et al. Results have been published, which show that the (111) surface has the highest hardness value ( Zhang et al., 2008b). 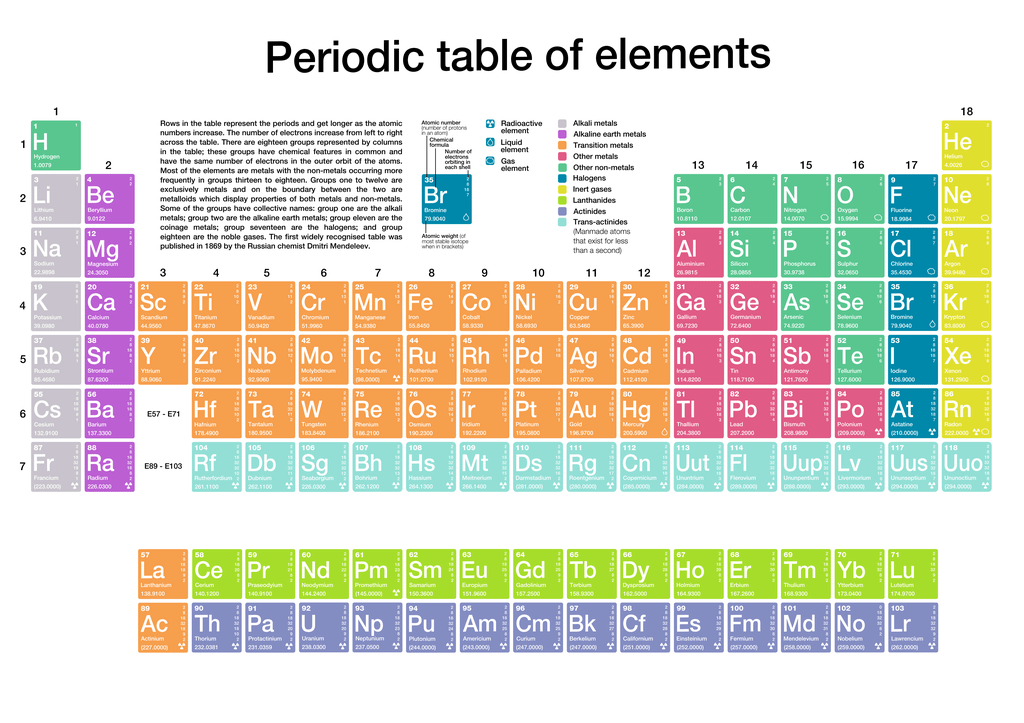
In general, the energy of defect formation increases with the addition of Zn ( Owens et al., 2002). The incorporation of Zn into the CdTe structure strengthens the crystal lattice ( Szeles, 2004), and considering that the Zn–Te ionicity is smaller than the Cd–Te ionicity, but the Zn–Te binding energy is higher, the stability of the compound increases as well as the shear modulus, while the densities of dislocations and subgrains are reduced ( Triboulet, 2005). This property leads to higher collection efficiency ( Duff et al., 2008) as the interactions between the crystal lattice and incoming radiation are increased. Although there is some discrepancy among authors, values between 5.8 and 6.68 g cc −1 ( Lun et al., 2006 Mandal et al., 2007) have been reported for CZT. CZT materials have a density of nearly 6 g cc −1 ( Auricchio et al., 2008 Fiederle et al., 2004 Goswami et al., 2005 Horodysky et al., 2006 Mandal et al., 2007 Marchini et al., 2008), which provides greater stopping power against incoming γ-rays than competing material technologies. One advantage of using CZT as a detector for high-energy radiation is the high average atomic number of Z = 50, compared with other well-known detector materials such as silicon and germanium with Z = 14 and Z = 32, respectively.ĭensity. Diéguez, in Comprehensive Semiconductor Science and Technology, 2011 3.04.2.2.1 Structural propertiesĪtomic number. 
If you have any doubts please reach out to us through the comments section, and we will get back to you as soon as possible.E. We hope this detailed article on Atomic Numbers will be helpful to you in your preparation. What happens if an atom loses a neutron?Īns: When you remove or add a neutron to the nucleus of an atom, the resulting substance is a new type of the same element and is called an isotope. Sometimes, adding a proton to an element makes the element radioactive. What happens if the number of protons is being changed?Īns: Changing the number of protons in an atom will change the atom from one element to a different element. The mass number of Phosphorus is \(31\) (Atomic Number Number of neutrons). The element with the atomic number \(15\) is Phosphorus. Which element has 15 electrons, 15 protons and 16 neutrons?Īns: If there are \(15\) protons in an atom, this means the atomic number of the given element is also \(15\). Lithium atoms contain \(3\) positively charged protons in their nucleus and \(3\) negative electrons outside the nucleus. Why does Z denote the atomic number?Īns: The atomic number is represented by the letter ‘Z’ because it is the conventional symbol that comes from the German word Zahl which means ‘number.’ Hence, Atomzahl means atomic number.Īns: Yes, a lithium atom is neutral because all atoms of a given element have the same number of protons and electrons given by the atomic number. We also learned how to calculate an atom’s neutron number and valency when its atomic number is given.įrequently Asked Questions (FAQs) on Atomic Number In this article, we learned the definition of atomic number, its representation, and the arrangement of elements in the Modern Periodic Table. Atomic Number represents the number of protons and electrons in a neutral atom the mass number represents the sum of protons and neutrons in a particular atom. 
However, the atomic number should not be confused with the mass number. It is the governing principle behind the arrangement of elements in the periodic table of elements. Each chemical element has a different number of protons, so the atomic number is a unique identifier for an element. The atomic number represented by Z refers to the number of protons in the nucleus of an atom and the number of electrons that surround the atom’s nucleus. Elements with similar chemical properties line up in the same column (group) ascending order of atomic numbers in the modern periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
